
ANRS MIE & DZIF Joint symposium at ESCMID Global 2026
ANRS MIE and DZIF organised the symposium ‘Shaping the Future of West Nile Virus Research in Europe’ as part of ESCMID Global 2026 in Munich
Last updated on 30 April 2026
In brief
- West Nile virus (WNV) infection is an endemic mosquito-borne zoonosis in Europe. The disease affects countries in Southern, Eastern and Western Europe
- ANRS MIE and DZIF are organising the symposium ‘Shaping the Future of West Nile Virus Research in Europe’ as part of ESCMID Global 2026 in Munich
The epidemiologic context
The West Nile virus (WNV) causes West Nile virus fever, a zoonotic disease. It was first isolated in 1937 in a woman in the Uganda Protectorate (now the Republic of Uganda), in the West Nile Province.
It is thought that the virus was introduced into Europe in the 1960s. France was the first European country to report human infections with WNV between 1962 and 1964.
Following a few sporadic outbreaks, the first major outbreak in Europe occurred in 1996 in Romania. Since 2010, outbreaks and epizootic foci have increased, particularly in Central Europe and Eastern Mediterranean, where the virus is endemic.
2018 was particularly notable, with the largest outbreak ever recorded in Europe. Human cases were reported for the first time in Germany in 2019 and 2020, and in the Netherlands in 2020.
In 2024, 19 European countries reported locally acquired human cases of WNV.

ANRS MIE & DZIF Joint Symposium
On the occasion of ESCMID Global 2026, ANRS MIE and the German Center for Infection Research (DZIF) organised a symposium dedicated to the future of West Nile virus (WNV) research in Europe.
The session was co-chaired by Marylyn ADDO, University Medical Center Hamburg-Eppendorf (UKE), German Center for Infection Research (DZIF), Germany; Eric D’ORTENZIO, ANRS Emerging infectious diseases, France; Anna PAPA-KONIDARI, Aristotle university school of medicine, Greece.
Objectives
The symposium brought together experts in virology, entomology, ecology and climate research, clinical research and public health, with the overall aim of establishing a joint research agenda capable of supporting earlier detection, strengthening our understanding of the mechanisms involved, and improving epidemic preparedness in Europe.
Among the topics addressed were the development of aligned research strategies and comparative studies on vector-borne diseases.
The programme
The programme consisted of four sessions:
- Arboviruses in Europe: framing West Nile virus within an evolving epidemiological and preparedness landscape. Marion KOOPMANS, Erasmus MC, the Netherlands
- Paris under threat: a One Health investigation of the 2025 West Nile virus emergence. Laura PEZZI, National Reference Centre (NRC) for Arboviruses, France
- Emergence and establishment of WNV in Germany. Christian DROSTEN, Institute of Virology, Charite Universitätsmedizin, Germany
- Clinical aspects and management of neuroinvasive West Nile virus infection. Luisa BARZON, EU Reference Laboratory for Public Health on Vector-borne Viruses, Italy
A brief overview
Arboviruses in Europe: framing West Nile virus within an evolving epidemiological and preparedness landscape
Marion Koopmans studies viruses at the human-animal interface and the processes that drive disease emergence at Erasmus MC, the Netherlands.

Marion Koopmans’ presentation set the stage for the symposium. It provided an overview of the current state of the art, knowledge gaps and ongoing research on drivers of emergence, spread and impact of West Nile virus outbreaks.
Transmitted mainly by female Culex mosquitoes, WNV is currently found in Africa, the Middle East, North America, Western Asia and Europe. Birds are the main reservoir of the virus: migratory birds from Africa introduced it into southern and western Europe, while local European birds can carry the virus during the winter. Humans and horses are dead-end hosts.
Marion Koopmans reminds us that in the Netherlands, the first autochthonous cases of infection identified in animals and humans were detected for the first time in 2020. This identification is the result of multidisciplinary surveillance and research activities on the virus focusing on mosquitoes, birds, horses and humans. The first locally acquired human case was identified 35 days after the discovery of the infected bird. Since then, between 2021 and 2023, research projects in animals revealed persistent local enzootic WNV circulation.
Climate change, amongst other factors, and its consequences (such as heavy rainfall and higher temperatures), as well as irrigation or stagnant water bodies, favour West Nile virus spread.
At the end of her presentation, Marion Koopmans concluded that monitoring WNV hosts and vectors could contribute to early detection of WNV circulation, and that such early detection could facilitate timely public health responses and allow more time to raise awareness among healthcare professionals.
Paris under threat: a One Health investigation of the 2025 West Nile virus emergence
Laura Pezzi is a researcher at the National Reference Centre (CNR) for Arboviruses in Marseilles. Her research focuses on the diagnosis, detection and characterisation of viruses in humans and mosquitoes.
In July 2025, the West Nile virus (WNV) emerged in Paris and northern France, with the first human cases ever recorded. The Île-de-France episode affected two patients with no history or risk factors, both of whom developed a neurological form of the disease.
Using samples collected as part of surveillance of humans, animals and mosquitoes between 2022 and 2025, Laura Pezzi and her team traced the spatial and temporal dynamics of the virus’s circulation in France, thereby characterising the lineage responsible for this emergence and identifying the most likely source of importation.
Only lineage 2 was detected. Phylogenetic analysis linked Mediterranean viral strains to those that had circulated in the Nouvelle-Aquitaine region (South Atlantic) in previous years. It also revealed that the 2025 outbreak in Paris was caused by viral strains also originating from the ‘South Atlantic region’ sublineage.
Historically, West Nile virus has been reported in the eastern Mediterranean region, comprising most of the Provence-Alpes-Côte d’Azur region and Corsica (1), and in the western Mediterranean region, including the coast of Occitanie (2), connected by the Camargue wetland, which overlaps with both regions. The most recent detections have occured in the South Atlantic area, specifically coastal Nouvelle-Aquitaine (3) and, in 2025, in the Paris region (4).
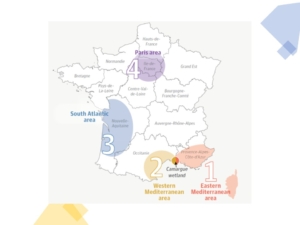
West Nile virus presence in France in 2025 (enomic epidemiology of West Nile virus in Paris. Klitting R, et al. JAMA Network Open. 2026;9(2):e2559588)
According to Laura Pezzi, given that WNV circulation in Europe is driven by several distinct mechanisms that cannot be determined on the basis of virus occurence data alone, genomic data are essential for establishing a link between viral strains circulating across regions.
She supports the ‘One Health’ approach, which relies on assessing virus spread by using samples from various components of the transmission chain (human, equine avian, and mosquitoe hosts). Cross-sectoral data integration, shared alert thresholds, and coordinated outbreak investigations are essential for better studying emerging zoonoses in Europe and responding to them effectively.
Emergence and establishment of WNV in Germany
Christian Drosten, director of the Institute of Virology at Charité Hospital in Berlin, specialises in the ecology, epidemiology and evolution of emerging viruses. He co-discovered SARS-CoV in 2003 and, together with his team, developed the first RT-PCR screening test in 2020, which is recommended by the WHO. He has also contributed to our understanding of MERS-CoV.

Christian Drosten presented a summary of the DZIF’s work on West Nile virus in Germany.
The virus was first detected in Berlin in 2018 in birds and in humans from 2019 onwards. The DZIF teams were able to identify and genetically characterise WNV infections in humans and mosquitoes, the vast majority of which were Culex pipiens mosquitoes. Viral sequences from human and mosquito cases were highly similar. The results showed that WNV lineage 2 infections were autochthonous and the virus had established an endemic maintenance cycle.
According to Christian Drosten, West Nile virus infections and associated symptoms, including neurological complications, are expected to increase in Berlin in the upcoming years. As observed in Italy and Spain, an increase in hot, prolonged summers in Germany, combined with worsening drought in Berlin and eastern Germany, is expected to increase West Nile virus transmission and associated infections.
Christian Drosten emphasises that the detection of patients with West Nile fever and the local maintenace of the virus in mosquitoes suggest that cases are under-diagnosed. Enhanced surveillance, combined with vector control in at-risk areas and efforts to raise awareness among physicians and vets are needed to monitor and limit WNV cases.
Clinical aspects and management of neuroinvasive West Nile virus infection
Luisa Barzon is affiliated with the EU Reference Laboratory for Public Health on Vector-Borne Viruses in Italy. Her clinical and research activities focus primarily on the epidemiology, genetics, pathogenesis and diagnosis of emerging pathogens, in vitro modelling of diseases, innate and adaptive immune responses to viral infections and vaccines, and the application of new biotechnologies in diagnostic microbiology.

West Nile virus (WNV) is transmitted by mosquito vectors between wild and domestic birds, which act as amplifying/reservoir hosts. Humans and equines are incidentally infected through Culex mosquitoe bite and may develop severe neurological disease; however, they are dead-end hosts, as the low level of viraemia cannot sustain onward transmission.
Infection in humans is generally asymptomatic. Fewer than 1% of patients develop neurological complications (meningitis, encephalitis, flaccid paralysis), with a mortality rate of between 10 and 20% among severe cases.
Luisa Barzon told us that in Italy, the regions reporting most human cases of infection were Veneto, Lombardy, Emilia-Romagna, Piedmont and Sardinia.
She presented the results of entomological, veterinary and human surveillance carried out in Veneto in 2022 and 2023 for WNV and the Ussuri virus (USUV), an RNA virus belonging, like WNV, to the genus Orthoflavivirus. Virological and/or serological analyses showed high viral circulation in the region during these two years. In 2022, early viral circulation was associated with earlier infection in wild birds and precocious occurence of a high number of human and equine WNV cases. This year, the endemic WNV lineage 2 was circulating alongside lineage 1, which had recently arrived in the region. Resident birds had a higher positivity rate than migratory birds. In 2023, the results were the reverse.
In 2023, viral circulation was lower than in 2022, as were infection rates in mosquitoes and birds and the number of human cases. There is currently no curative treatment for humans. In cases of neuroinvasive forms, patients receive supportive care, involving hospitalisation, respiratory support and close monitoring of vital signs.
In Europe, a drier winter, high spring temperatures and summer drought are associated with earlier and more widespread circulation of WNV in vectors, birds and humans.
For Luisa Barzon, the results obtained in Veneto support the efficacy of the ‘One Health’ approach for WNV surveillance. In this approach, human medicine, entomology and veterinary disciplines are interconnected and mutually functional.
This joint ANRS MIE/DZIF symposium highlighted the urgent need to expand and coordinate genomic surveillance accross Europe, particularly in historically endemic regions and areas recently affected. During the discussion following the presentations by the various speakers, it was emphasised that there is currently no curative treatment available for humans. In terms of prevention, no vaccines are yet available either. However, vaccines that have proven effective in horses could serve as models for the development of human vaccines.
Bibliography
- A multidisciplinary approach to the detection of and response to West Nile virus in the Netherlands between 2020 and 2023: best practices, challenges and opportunities
- Genomic Epidemiology of West Nile Virus in Paris
- Ecological and clinical evidence of the establishment of West Nile virus in a large urban area in Europe, Berlin, Germany, 2021 to 2022
- Integrated One Health Surveillance of West Nile Virus and Usutu Virus in the Veneto Region, Northeastern Italy, from 2022 to 2023

ANRS MIE / SFV “Emerging Viruses” 2025 Thesis Awards
In April 2026, the ANRS MIE and the SFV awarded three thesis prizes to Morena Gasparine, Riccardo Vernuccio and Konrad M. Wesselmann.
18 May 2026



